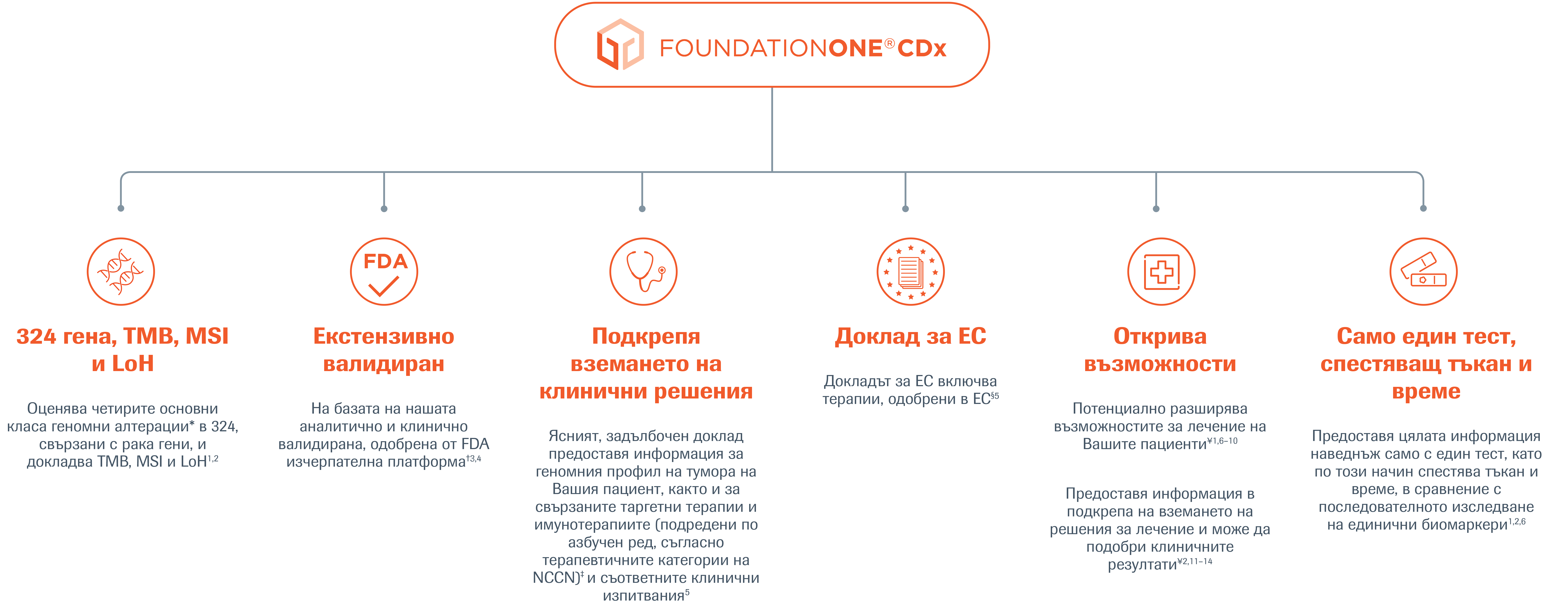
Нашата иновативна, валидирана от FDA тъканно-базирана услуга за изчерпателно геномно профилиране за всички солидни тумори, която помага за вземането на ефективни, персонализирани решения за лечение. 1─3

Референции:
1. Frampton GM et al. Nat Biotechnol 2013; 31: 1023–1031.
2. FoundationOne®CDx Technical Specifications 2020. Available at: https://www.foundationmedicine.com/test/foundationone-cdx (Accessed May 2021).
3. FoundationOne®CDx FDA Approval, 2017. Available at: https://www.fda.gov/medical-devices/recently-approved-devices/foundationone-cdx-p170019 (Accessed May 2021).
4. FoundationOne®CDx Clinical and analytical validation, https://www.rochefoundationmedicine.com/content/websites/fmi-landing/en/accessandfunding.html (Accessed May 2021)
5. FoundationOne CDx Sample Report. Available at: https://assets.ctfassets.net/w98cd481qyp0/2pusoGEB1tZXWHYL3vlDnv/b2a6ede771218a007f9ca36fe76c2c70/F1CDx_NSCLC_CDx_Elevated_TMB.pdf (Accessed May 2021).
6. Drilon A et al. Clin Cancer Res 2015; 21: 3631–3639.
7. Rankin A et al. Oncologist 2016; 21: 1306–1314.
8. Ross JS et al. Cancer 2016; 122: 2654–2662.
9. Suh JH et al. Oncologist 2016; 21: 684–691.
10. Hirshfield KM et al. Oncologist 2016; 21: 1315–1325.
11. Rozenblum AB et al. J Thorac Oncol 2017; 12: 258–268.
12. Schwaederle M et al. Mol Cancer Ther 2016; 15: 743–752.
13. Wheler JJ et al. Cancer Res 2016; 76: 3690–3701.
14. Dhir M et al. Cancer Med 2017; 6: 195–206.
15. Zhao P et al. J Hematol Oncol 2019; 12: 54.
16. Abida W et al. JAMA Oncol 2019; 5: 471–478
17. FDA approves pembrolizumab for first-line treatment of MSI-H/dMMR colorectal cancer. Available at: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-pembrolizumab-first-line-treatment-msi-hdmmr-colorectal-cancer (Accessed May 2021).
18. NCCN Clinical Practice Guidelines in Oncology. Prostate Cancer. Version 2.2021, Feb 17 2021. Available at: https://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf
19. Kok M et al. ESMO Open 2019; 4(Suppl 2): e000511.
20. Gandara DR et al. Nat Med 2018; 24: 1441–1448.
21. FDA approves pembrolizumab for adults and children with TMB-H solid tumors, 2020. Available at: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-pembrolizumab-adults-and-children-tmb-h-solid-tumors (Accessed May 2021).
22. Marabelle A et al. Ann Oncol. 2019;30(suppl_5):v475-v532.
23. Yarchoan M et al. JCI Insight 2019; 4: e126908.
24. Swisher EM et al. Lancet Oncol 2017; 18:75−87.
25. Coleman RL et al. Lancet 2017; 390:1949−61.
26. Merker JD et al. J Clin Oncol 2018; 36: 1631–1641.
27. Scheerens H et al. Clin Transl Sci 2017; 10: 84–92.
28. NCCN Clinical Practice Guidelines in Oncology. Non-Small Cell Lung Cancer. Version 4.2021- March 3, 2021. Available at: https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf (Accessed May 2021)
29. Schrock AB et al. Clin Cancer Res 2016; 22: 3281–3285.
30. Ali SM et al. Oncologist 2016; 6: 762–770.
31. Pekar-Zlotin M et al. Oncologist 2015; 20: 316–322
32. FoundationOne Liquid CDx Technical Specifications, 2020. Available at: https://www.foundationmedicine.com/test/foundationone-liquid-cdx (Accessed May 2021).
33. Woodhouse R et al. (2020) Clinical and analytical validation of FoundationOne Liquid CDx, a novel 324-Gene cfDNA-based comprehensive genomic profiling assay for cancers of solid tumor origin. PLoS ONE 15(9): e0237802. September 25, 2020 Accessed at: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0237802 (Accessed May 2021).